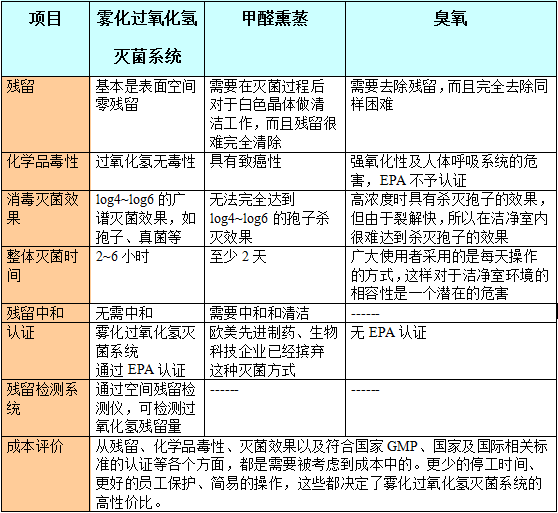
The goal of GMP for pharmaceutical companies is to ensure the establishment of a scientific and rigorous sterile pharmaceutical production environment, process, operation and management system to minimize all possible and potential biological activity, dust, pyrogen pollution and produce high quality. , hygienic and safe pharmaceutical products. This provides a so-called "sterile" environment. First of all, we need to understand the types and differences of pharmaceutical plants. The pharmaceutical factories in China can be divided into six types, namely antibiotics factories, chemical synthetic pharmaceutical factories, preparation plants, biochemical pharmaceutical factories, by-product drugs factories, and Chinese medicine factories. and many more. Then a drug, reagent, prototype, etc. produced by a pharmaceutical factory does not have the safety limit of the hard indicators of the air quality in the production workshop. It is used in the market to use the people. This series of chain reactions is incredible! So what is the air purification in the workshop of the pharmaceutical factory? The main purpose of the air purification system of the pharmaceutical factory is to prevent microbial contamination of products and clean areas, to prevent the spread and pollution of viruses, pathogenic bacteria and spores used in pharmaceutical production, to prevent the spread and pollution of penicillin, and to prevent the spread of solid dust. . As the top priority of GMP, the air quality provided by the pharmaceutical GMP workshop will directly affect the level of particulate and microbial contamination of the drug produced in the environment, which will directly affect the quality of pharmaceutical production. Therefore, air is the main medium for invading the pollutants of our products. To ensure the stability and reliability of the cleaning and disinfection effect, it is very important for pharmaceutical manufacturers to establish appropriate cleaning and disinfection standard operating procedures and guide operators to master effective cleaning and disinfection methods. At present, the most common environmental disinfection methods used by pharmaceutical companies are formaldehyde and ozone. However, with the advancement of society, people are paying more and more attention to the adverse effects of such gas disinfectants. The Ministry of Health's 2000 and 2002 editions of the "Disinfection Technical Specifications" all proposed "formaldehyde carcinogenicity, not suitable for indoor air disinfection." Ozone promotes rubber seal aging, its residual concentration requirement is less than 0.16mg/m 3 , at this lower limit, Difficult to test and control. Ozone disinfection is inadequate for aseptic processing areas. Work clothes that are operated in non-sterile areas are over-sterilized by ozone, which also has an adverse effect on clean clothes, causing defects such as easy-to-drop fibers. After the new standard is improved, the disinfection cannot be blindly emphasized and its negative effects are ignored. Sterilization of GMP workshops in pharmaceutical factories is a key point and a difficult point. At present, most of the pharmaceutical factories use formaldehyde fumigation sterilization, but the strong toxicity residue of formaldehyde fumigation will exist in the clean area for a long time, which has a long-term potential for operators and the environment. Hazard; For the health and production management needs of employees, QA has gradually realized the importance and necessity of finding a new GMP sterilization product to replace formaldehyde fumigation. Â Â Â Â Hydrogen peroxide sterilization is the ideal space for disinfection and sterilization of pharmaceutical factories Hydrogen peroxide space sterilization technology is a highly efficient, non-toxic, residue-free sterilization method. It has been widely used in the environmental sterilization of the pharmaceutical and biosafety industries, replacing traditional UV lamps or ozone, formaldehyde fumigation, etc. Bacterial method. Its main advantages are: high sterilization efficiency, absolute safety for personnel, and zero residue to the environment, which is an ideal alternative technology for environmental disinfection and sterilization. Metso dry mist hydrogen peroxide sterilizer has various shapes, simple operation, convenient carrying and good atomization performance. Using compressed air as the carrier gas, the disinfectant is supplied quantitatively. When a small amount of liquid enters the nozzle through the siphon principle, it encounters a high-speed carrier gas, and the gas strongly collides and pulverizes the droplet, and rapidly transports the dry mist particles below 10 microns. Spread to all locations in the clean area to the distal end for efficient sterilization. It is disinfected and sterilized at log6 level with a special low-concentration hydrogen peroxide sporicide, and it is not corrosive to articles and color steel plates in the environment. Metso dry mist hydrogen peroxide sterilizer has the following advantages over traditional sterilization methods in GMP clean room sterilization: 1. With efficient sterilization, this is currently beyond the reach of other traditional disinfection methods; 2. The surface space is basically zero residue and ecologically friendly; 3. New front-end technology, approved by microbiological testing of third-party hospitals and medical institutions. 4. Good trial experience, light and easy to move, ergonomic design; 5. Integrated three-dimensional disinfection, one device can effectively sterilize the environment, voids, surface, 360 degrees without gaps, no dead ends; 6. Fully automatic control, saving a lot of labor costs and avoiding disinfection failure caused by human factors; Metso dry mist hydrogen peroxide sterilizer has been successfully applied to the highest disinfection standard places such as hospital wards, nursing homes, pharmaceutical clean rooms, sterile scientific laboratories, etc., and has achieved excellent results and users' praise. summer garment,autumn garment,winter garment Ningbo Staneex Imp. & Exp. Co., Ltd. , https://www.staneex.com